Scientific description of Potato Wart Disease (2020)
Based on Adolf B. et al. (2020) Fungal, Oomycete, and Plasmodiophorid Diseases of Potato. In: Campos H., Ortiz O. (eds) The Potato Crop. Springer, ChamThe author of this content is Jaroslaw Przetakiewicz
The pathogen causing potato wart disease, Synchytrium endobioticum (Schilb.) Perc., was first discovered by Schilberszky in Hungary. In fact, the pathogen was known firstly in Europe.
In 1876, potato wart disease was found for the first time in the UK (Hampson 1993; Flath et al. 2014). In the older classification this species has been included to Protista Kingdom. Nowadays, Synchytrium endobioticum (Schilb.) Perc., belongs to Fungi Kingdom, phylum Chytridiomycota, order Chytridiales, family Synchytriaceae, genus Synchytrium, and species endobioticum. The genus Synchytrium included about 200 species which are endobiotic halocarpic organisms that have inoperculate sporangia. All species of the genus Synchytrium are parasites but the most important economically and phytosanitary is S. endobioticum the causal agent of potato wart disease.
The disease is also known by various common names like black wart, cauliflower disease, warty disease, potato tumor, potato cancer, black cancer, or black scab.
S. endobioticum is an obligate soil-borne biotrophic fungus which is considered to be the most important worldwide quarantine plant pathogen of cultivated potato. Cultivated potato (Solanum tuberosum) is the primary host, but the fungus, under experimental conditions, can also infect wild species in genera Capsicastrum, Duboisia, Hyoscyamus, Lycium, Nicotiana, Nicandria, and Physalis (Obidiegwu et al. 2014).
The pathogen is a primitive fungus which stimulates its host to produce hypertrophic outgrowths on young potato organs, such as eyes, sprouts, young tubers, stolons, stems, leaves, and even flowers but never roots. The fungus does not form hyphae but forms sporangia that produce about 200–300 motile zoospores (Obidiegwu et al. 2014).
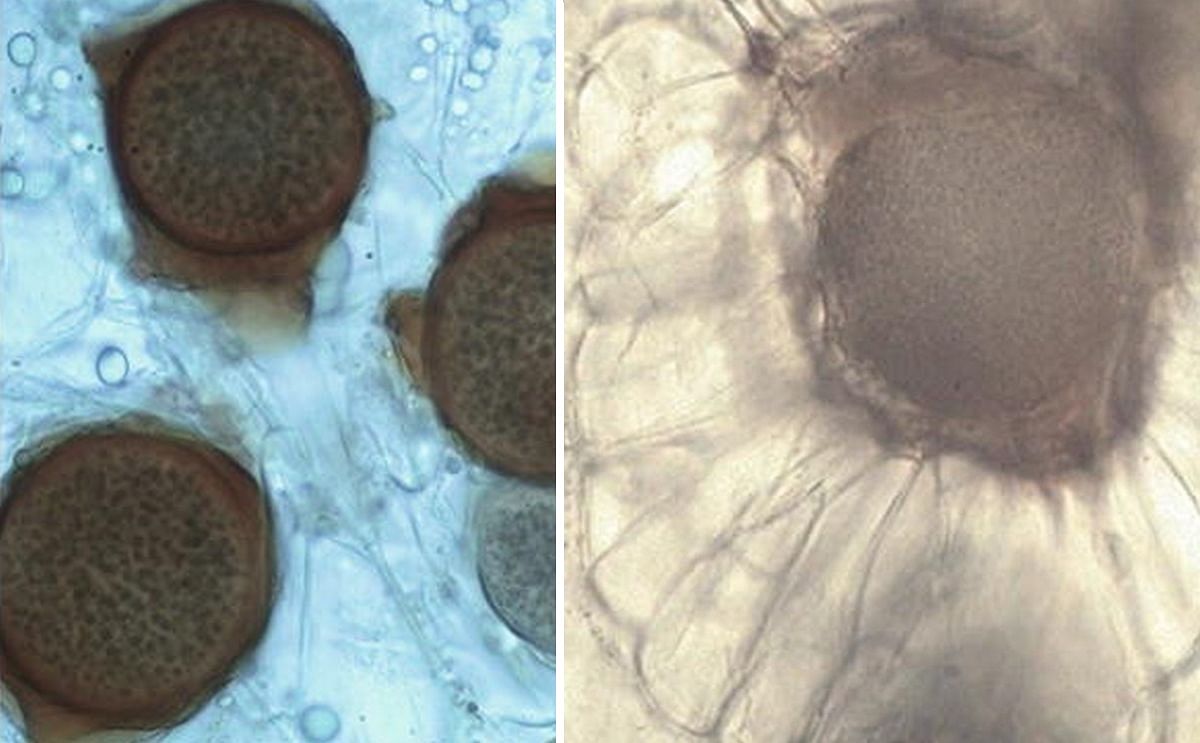
Summer (left) and winter (resting) sporangia (right) of Synchytrium endobioticum. Thin-walled summer sporangia of pathotype 3(M1) are enclosed within one membrane forming sorus on surface of wart tissue of cultivar Asche Sämling. Thick-walled winter sporangia of pathotype 8(F1) in wart tissue of cultivar Sonda.
- Summer sporangia (image above) have a thin cell wall and form haploid zoospores which are emerging and steady reinfection of the host tissue like sprouts, tubers, eye tubers, stolons, and roots (only in tomato) (Przetakiewicz 2014a). In appropriate conditions after isogamy of haploid zoospores to diploid zygotes which are able to infect host cells and form winter sporangia which are embedded deeper into the host tissue than the sori (always on the surface).
- Winter (resting) sporangia (image above) are the dormant structures by which the fungus disperses to establish new infections. They are usually spherical to ovoid in shape and 24–75 μm in diameter with thick-walled (triple wall) structure, which is ornamented with irregularly shaped wing-like protrusions. The spores can survive for a long time without plant hosts. After 43 years, in favorable conditions, disease may develop even from single spores of S. endobioticum (Przetakiewicz 2015b, 2016).
Similar diseases
Simultaneous germination of all buds in one eye results in wart-like outgrowths very similar to those caused by S. endobioticum. However, these pseudo-warts consist of abundant pointed shoots compacted together. No winter sporangia are present in the affected tissue.Symptoms of powdery scab caused by Spongospora subterranea f.sp. subterranea or potato smut caused by Thecaphora solani can be mistaken for wart occurrence. A view under the microscope reveals that spore balls look different from winter sporangia of S. endobioticum.
Impact of Potato Wart disease
Potato wart disease is so important that, for some 65 years, quarantine and domestic legislations have been in force throughout the world to prevent its spread (Anon 2015). The economic impact of disease caused by this pathogen is not only from disease losses but from loss of international trade markets, long-term quarantines, and regulatory restrictions placed on infested areas and the buffer zones (Przetakiewicz 2014a).
Chemical control of S. endobioticum is not possible. The only strategies to confine the disease are strict quarantine and phytosanitary measures as well as cultivation of resistant cultivars (Obidiegwu et al. 2014). The availability of resistance cultivars allowed governments to issue regulations prohibiting the cultivation of susceptible cultivars (Baayen et al. 2006). For example, since 1955 only cultivars resistant to pathotype 1(D1) could be registered and grown on the Polish territory. As stated in the Food and Veterinary Office Mission Report, no potato plants or tubers with symptoms of S. endobioticum pathotype 1(D1) have been detected in Poland since the 1950s–early 1960s. This is a result of growing only resistant cultivars to mentioned pathotype of S. endobioticum (Przetakiewicz 2008).
Worldwide prevention is based on the control of disease spread and pathogen exclusion via regulatory action. S. endobioticum has a very limited capacity for natural spread, which is principally why it has been possible to control it so effectively by statutory means. Nevertheless, S. endobioticum is a classic example of the distribution of plant pathogens by man. Regulatory action has largely restricted the spread of the disease within potato-growing regions, as the seriousness of the disease was quickly recognized (Hampson 1993). Once S. endobioticum has been introduced into a field, the whole crop may be rendered unmarketable and moreover the fungus is so persistent that potatoes cannot be grown again safely for many years, nor can the land be used for any plants intended for export.
Based on the results obtained by Przetakiewicz (2015b), winter sporangia of S. endobioticum are very persistent and capable of retaining viability for as long as 46 years. The single spores from the inter-host period were still infective after 43 years. These results should be taken into consideration when de-scheduling previously infested plots even after 40 years or longer, especially in the mountainous areas. S. endobioticum is still of great economic importance in cool areas and wet mountainous regions.
The detection of potato wart disease on Prince Edward Island during the 2000 growing season resulted in an estimated $30 million loss to the island’s economy in the first year.
Pathogen Populations and Distribution
Potato wart disease appears to have arisen in the potato-growing area of Andean South America (Hampson 1993). Wart-like outgrowths on early Peruvian tuber-shaped pottery were interpreted as potato wart disease. The disease is likely to have arrived in Europe from South America because the Great Potato famine of the 1840s in Ireland induced European growers to import potato germplasm from South America. The introduction of the pathogen to Europe was possible by diseased tubers, infested soil or contaminated bags along with shipments of guano (Hampson 1993). S. endobioticum spread at the end of the nineteenth century from the center of origin in the Andes first to Europe and North America and subsequently across whole potato-growing areas of Asia, Africa and Oceania. Historic account has it that potato wart disease entered England in 1876 or 1878 while another view upholds that the disease has been present in the Liverpool province of England in 1876 or 1878 (Obidiegwu et al. 2014). In 1901, the disease was officially recognized in the UK. Potato wart disease and its causal agent were described by Schilberszky who received in 1888 warted tubers of cultivar Maercker-Zwiebel that had been grown locally from seed tubers imported from England. It spread widely in Europe, but statutory measures finally restricted its distribution and it has spread only to a limited extent to other parts of the world.
According to EPPO Pest Quarantine Database (Anon 2015), S. endobioticum occurs locally in almost all EPPO countries. The distributions are fragmentary as a result of statutory control. According to national reports, it has been found but is not established in Belgium, France, Luxembourg as well as in Lebanon. Found in the past but eradicated in Portugal (unconfirmed). In Asia countries S. endobioticum occurs in Armenia, Bhutan, China, India, Georgia, Nepal, Turkey. In Africa: South Africa, Tunisia, Algeria, Zimbabwe, and Egypt (absent, unreliable record). North America: Canada, Mexico (absent formerly present), USA (eradicated). South America: Bolivia, Chile (eradicated), Ecuador, Falkland Islands, Peru, Uruguay (absent, confirmed by survey). Oceania: New Zealand (South Island).
Numerous pathotypes of the fungus occur and are defined by their virulence on differential potato cultivars. In Europe, more than 40 pathotypes of S. endobioticum have already been identified (Obidiegwu et al. 2014; Przetakiewicz 2014b, 2015a). A pathotype is characterized by its pattern of virulence or avirulence to a series of differential cultivars of potato. Pathotypes 1(D1), 2(G1), 6(O1), 8(F1), and 18(T1) are the most relevant in Europe. Other pathotypes occur mainly in the rainy mountainous areas of central and eastern Europe (Alps, Carpathians). They persist mainly in small garden potato plots and not in commercial potato crops (Przetakiewicz 2014a). Mitochondrial genomic variation shows that S. endobioticum has been introduced into Europe multiple times, that several pathotypes emerged multiple times, and that isolates represent communities of different genotypes (van de Vossenberg et al. 2018a).
Resistance
The biggest discovery was finding resistant cultivars of potato to S. endobioticum among cultivated ones.
Systematic studies on resistance of potato cultivars to S. endobioticum started in England in 1909. Resistance sources to S. endobioticum were found in old cultivars such as Snowdrop and Flourball, which facilitated resistance breeding. Conventional breeding programs were successful in controlling potato wart disease through the development of resistant cultivars early in the twentieth century (Obidiegwu et al. 2014). Breeding for resistance was successful, thanks to the arability of a dominant gene that blocked development and reproduction of originally introduced pathotype 1(D1) of S. endobioticum.
Unfortunately, in Europe wart development on resistant potato cultivars was first discovered in 1941 in Germany [pathotype 2(G1)] and former Czechoslovakia [pathotype 3(S1)]. The new pathotypes have been proved to be difficult to control and eradicate then the original pathotype 1(D1) (Baayen et al. 2006). Molecular mapping studies provide evidence that wart resistance to pathotype 1(D1) of S. endobioticum can be conferred by a single locus from different sources. Hehl et al. (1999) mapped the single dominant gene Sen 1 for resistance to pathotype 1(D1) in diploid mapping population. The gene Sen 1 is located on potato chromosome IX. Brugmans et al. (2006) also used a diploid potato linkage map to locate Sen1-4, a second dominant gene for resistance to pathotype 1(D1). This gene is located on the long arm of chromosome IV. In these two mentioned populations, the resistance segregated as a monogenic trait. Ballvora et al. (2011) discovered the first loci for virulent pathotypes 2(G1), 6(O1), and 18(T1) in two tetraploid half-sib families, in which the resistance to pathotype 1(D1) also segregated. In contrary to earlier studies in diploid populations (Hehl et al. 1999; Brugmans et al. 2006), the phenotypic distribution of wart resistance appeared quantitative in the two mapping populations analyzed (Ballvora et al. 2011). The quantitative resistance locus (QRL) Sen2/6/18 on chromosome I expressed resistance to pathotypes 2(G1), 6(O1), and 18(T1). The QRL Sen18 on chromosome IX expressed resistance to pathotype 18(T1). And the third QRL Sen1 on chromosome XI expressed resistance mainly to pathotype 1(D1) (Ballvora et al. 2011). Groth et al. (2013) mapped quantitative trait loci (QTL) for resistance to pathotype 1(D1), 2(G1), 6(O1), and 18(T1). The QRL for all four pathotypes were located on chromosomes II, VI, VIII, and IX, and QRL for pathotypes 2(G1), 6(O1), and 18(T1) on chromosomes VII and X. The QRL detected in this study were different from the ones in Ballvora et al. (2011). The cultivar Panda used in this study (Groth et al. 2013) as a resistant parent to pathotype 1(D1), 2(G1), 6(O1) and 18(T1), is in fact slightly susceptible to all four pathotypes. These results were confirmed in CORNET project (acronym SynTest) (Przetakiewicz, unpublished results 2014) Consequently, breeding is hampered by a lack of dominant major genes for resistance to virulent pathotypes. The wart resistance has several sources and diverse backgrounds which resulted in a few potato cultivars resistant to virulent pathotypes of S. endobioticum. Nevertheless, recent results indicated for identify a newly locus Sen2 located on chromosome XI which provides resistance to at least seven various virulent pathotypes of S. endobioticum (Plich et al. 2018).
Management
The first acts were issued in 1908 in Ireland, and a few years later in Scotland, England, and Germany. The first legislation related primarily to prohibit the import from other countries of potatoes infected by S. endobioticum.
In that time the acts did not take into account the possibility of the spread of the disease within the country. It was a reason of increasing number of outbreaks in such a short time in many countries. In consequence of the wide spread of potato wart disease was the development of research on the biology controlling of the fungus.
EPPO includes S. endobioticum into the A2 quarantine list because of its long persistence in soil and the severe losses it inflicts to potato crops. The United State Department of Agricultural (USDA) has the fungus on its official list of selected agents and toxin. The European Union issued a specific requirement in the Council Directive 69/29/EC of 8 December 1969 on control of Potato wart disease and the Council Directive 2000/29/EC of 8 May 2000 on protective measures against the introduction into the Community of organisms harmful to plant or plant products and against their spread within the Community (Obidiegwu et al. 2014).
Looking Forward
Although S. endobioticum seems to be in remission in Europe, new foci have appeared in countries with warmer continental climate (Turkey, Georgia, Bulgaria, and Greece) (Anon 2015; Gorgiladze et al. 2014; Vloutoglou pers comm. 2015).
This may suggest adaptation of S. endobioticum to warmer and/or dryer climate as well as the lack of adequate controls in countries where the disease has not been present before. Moreover, new pathotypes have been discovered recently (Çakir et al. 2009; Przetakiewicz 2015a). It can be expected that new pathotypes of S. endobioticum might appear, since there are many reasons for new pathotypes to arise (Melnik 1998).
The newest Pathotype 39(P1) was detected in the rainy mountainous area, in small garden potato plots, when the old traditional cultivars of potato are cultivated without any crop rotations. It seems therefore that where climatic conditions are suitable for S. endobioticum to take place and the growing of slightly susceptible cultivars is possible, the development of new pathotype is favored (Przetakiewicz 2015a).
Although, local pathotypes persist mainly in small garden potato plots in economically unimportant potato-growing regions, they are still very important for quarantine and phytosanitary measures as spores can spread in rain water (Przetakiewicz 2015b). Phenotypic assessment of resistance to S. endobioticum is laborious and, time-consuming.
Diagnostic DNA-based markers closely linked with or, even better, located within wart resistance genes would greatly facilitate the early detection and combination of different resistance sources and are therefore highly desirable (Obidiegwu et al. 2014). Natural DNA variation in wild and cultivated potato germplasm provides an excellent platform for the discovery of diagnostic tools for marker-assisted selection and resistance gene cloning (Obidiegwu et al. 2014). A new dominant gene Sen2 on chromosome XI provides extreme resistance to pathotypes 1(D1), 2(G1), 2(Ch1), 3(M1), 6(O1), 8(F1), 18(T1), and 39(P1). In the future, this gene will offer potentials for the efficient selection of new commercial cultivars that are resistant to multiple S. endobioticum pathotypes. EPPO Standard PM 7/28 (2) (Anon 2017) recommends various biotests using differential potato cultivars for the identification only 4 pathotypes [1(D1), 2(G1), 6(O1) and 18(T1)].
Pathotype determination is labor-intensive and time-consuming too, especially for the identification of local pathotypes which require more differential cultivars (Przetakiewicz 2017; Przetakiewicz and Plich 2017). Molecular diagnostic tools (TaqMan PCR method) are currently available for the identification of pathotype 1(D1) and its discrimination from non-1(D1) pathotypes (Bonants et al. 2015; van de Vossenberg et al. 2018b). However, this method requires costly probes and cannot be used for the identification of virulent pathotypes. The recent report revealed no sequence polymorphisms between the five S. endobioticum pathotypes, indicating that S. endobioticum pathotypes may be exceptionally similar to each other. One reason for this may be that the development of new S. endobioticum pathotypes is caused by very limited changes in avirulence factors rather than extensive genetic recombination between divergent genotypes (Busse et al. 2017). Recently, polymorphic microsatellite markers were used to assess the genetic diversity of potato wart at the intraspecific level for the first time and will certainly contribute to a better understanding of the evolutionary history of this pathogen in the years to come (Gagnon et al. 2016).
The molecular methods for S. endobioticum detection based on the RealTime PCR (Smith et al. 2014; Bonants et al. 2015) or PNA-based hybridization assay (Duy et al. 2015) have several advantages over the traditional method of microscopic examination in routine diagnostic testing. Molecular methods can significantly reduce the time to disease diagnosis and prevent the spread of S. endobioticum to other locations.
