Scientific description of Powdery Scab Disease (2020)
Introduction and Future PerspectivesSpongospora subterranea causes root hyperplasia (root galling) and tuber powdery scab, leading to losses in seed and ware crops of potato worldwide and is also important as the natural vector of Potato mop-top virus (PMTV), an economically important tuber blemish disease of potato found in some regions.
Powdery scab is particularly favored by cool, damp conditions and is an intractable disease. Additionally, S. subterranea is an unculturable biotroph, making research difficult. Progress in understanding many aspects of the biology of S. subterranea has been slow relative to other plant pathogens and even compared with other plasmodiophorids.
However, the development of quantitative molecular assays and the increasing availability of sequence information have allowed recent progress to be made in understanding various aspects of the epidemiology of powdery scab and root galling, and biology of the pathogen.
The pathogen can survive for many years and very low levels of inoculum can cause relatively severe disease outbreaks, making control elusive: it is generally accepted that an integrated approach to disease control will prove most effective as no single control method is totally effective.
Additional knowledge of inoculum- based risk, pathogen variation and infection conditions will all contribute towards such a risk-based integrated control system. The tripartite potato/S. subterranea /PMTV interaction is a difficult system to manipulate and there has been little progress in understanding the basis of host specificity and molecular mechanisms of virus transmission.
In recent years some progress has been made in developing molecular markers and genomic sequence information for the plasmodiophorids. In addition, host genomics has advanced with the discovery of genetic markers and identification of novel resistance.
The availability of in vivo root culture systems for S. subterranea propagation, high- throughput nucleotide sequencing, comparative genomics and advanced imaging technologies will no doubt be applied in due course in order to advance knowledge of this intractable and understudied disease and the virus that it vectors.
It is envisaged that availability of plasmodiophorid genomes will, in the future, lead to molecular interaction studies between S. subterranea and compatible host plants, and will allow the genes or molecular mechanisms involved in host recognition, infection of host cells, multiplication of S. subterranea within roots and the development of sporangial or sporogenic phases to be identified.
Symptoms
Spongospora subterranea can infect all underground organs of potato (i.e. stolons, tubers, and roots) where the pathogen stimulates the enlargement and division of host cells leading to the appearance of symptoms.
Depending on environmental conditions initial tuber symptoms, which take 4–8 weeks to develop, are purplish brown lesions (1–2 mm diameter) that subsequently enlarge into raised mature lesions which burst, exposing large masses of sporosori resulting in characteristic symptoms of the disease (Image below).

Characteristic lesions of powdery scab on cultivar Agria.
Infection of roots or stolons can sometimes lead to development of galls (hyperplasia) (image below), where infected tissue is stimulated to grow and sporosori are formed inside the gall (image below).
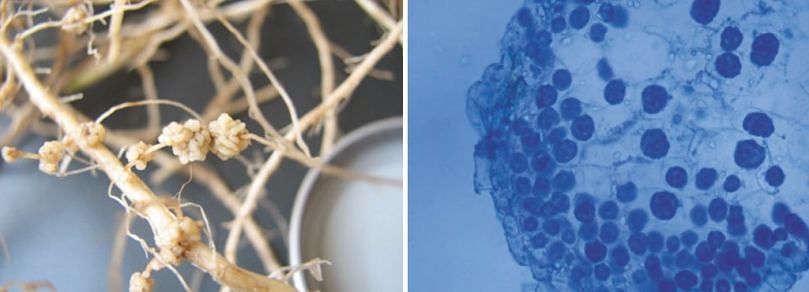
Root galling of potato caused by Spongospora subterranea (left), Sporosori inside root gall (right)
Impact
The main impact of powdery scab is cosmetic, due to a reduction in quality caused by lesions on the tuber surface, thus decreasing their value for either fresh or processing purposes. The disease also affects seed tuber certification, with the extent of the impact dependent on certification tolerances which vary from country to country (Falloon 2008; Wale 2000).
Reports of losses due to unmarketable tubers can be as high as 50% in Australia (Hughes 1980) to 100% of the harvested product in Venezuela (Bittara et al. 2009). In addition, root infection by S. subterranea can reduce water absorption and nutrient intake (Shah et al. 2012).
Some studies have reported a reduction in tuber yield and mean tuber weight due to disease caused by S. subterranea on either Solanum tuberosum spp. andigena or S. tuberosum spp. tuberosum (Gilchrist et al. 2011; Shah et al. 2012). In contrast, field studies using the cultivars Shepody and Umatilla Russet (S. tuberosum spp. tuberosum) showed no detrimental effect on either tuber yield or mean tuber weight due to the development of symptoms on roots and tubers (Johnson and Cummings 2015).
Spongospora subterranea is also the vector of Potato mop-top virus (PMTV), one of the causes of spraing, a tuber blemish disease (Jones and Harrison 1969).
PMTV is carried within zoospores and S. subterranea can remain viruliferous for many years. Little is known, however, about the virus–vector interactions, particularly with respect to the conditions conducive to transmission of the virus and the differential symptom expression of one or other of the diseases where infection by both organisms has occurred.
Pathogen Populations and Distribution
Since the first description of Spongospora subterranea under the name Erysibe subterranea by F. W. Wallroth in 1841, the pathogen has been reported in most potato- growing regions of the world, including hot and dry countries where farming is conducted at high altitudes or under irrigation. The number of first reports continues to increase across the world (Harrison et al. 1997; Merz and Falloon 2009; Wale 2000).
Few attempts have been made to characterize variation in S. subterranea, an unculturable biotroph with poorly understood genetics. It is difficult to obtain sufficient “clean” DNA for commonly used molecular marker techniques, sequence information is limited and assumptions about what constitutes an isolate or “strain” (sporosorus, resting spore or zoospore) are made.
It is not demonstrated that sporosori occur as a result of sexual recombination, although this is thought to be the case (Braselton 1995). In general, analyses have been made on a single sporosori, which may also comprise many genotypes in the form of individual resting spores.
Genetic variation in Internal Transcribed Spacer sequences revealed limited differences among collections from Europe and Australasia and those from South
America (Bulman and Marshall 1998). Three ribotypes (I, II, and III) of S. subterranea were identified, with ribotype I, II and III is found in South America, ribotype II in North America and Australasia and ribotypes I and II in Great Britain (Osorio- Giraldo et al. 2012; Qu and Christ 2004).
Most recently, Gau et al. (2013) applied SSR markers and ITS and actin sequence data to approximately 700 samples of S. subterranea obtained from 19 countries, different resting spore sources of the pathogen (root galls or tuber lesions) and from different potato host (sub) species.
They described an overall low genetic diversity in S. subterranea, comprising three sample clusters; two occurring in South America (root galls and tuber lesions) and the third group comprising samples from elsewhere, independent of the resting spore source, and suggested that an ecological adaption in the native region due to coevolutionary processes and/or competitive exclusion may have taken place.
South American populations were more diverse than those from other regions but no relationship between host species and pathogen diversity was noted.
Host Resistance
Resistance to powdery scab exists in some cultivars worldwide (Torres et al. 1995; Genet et al. 1996; Christ 1987; Falloon et al. 2003; Nitzan et al. 2008; Merz et al. 2012; Bittara et al. 2016). Falloon et al. (2003) found that although cultivars ranged from very susceptible to resistant, all developed zoosporangia and root galls and therefore none possessed immunity.
Susceptibility of individual cultivars to root and tuber infection is not always closely correlated (Hughes 1980; Falloon et al. 2003; Bittara et al. 2016), particularly with regards to the relationship between root and tuber infection and root galling (Falloon et al. 2003; van de Graaf et al. 2007).
The use of certain cultivars with high tuber resistance may therefore still maintain populations of S. subterranea in the soil. Despite the availability of disease resistant cultivars, genetic resistance to S. subterranea currently plays a minor role in disease control as cultivars are usually selected by growers for characteristics other than their ability to resist powdery scab.
Resistance to powdery scab has also been demonstrated in other Solanum species, for example, S. acaule was reported to show resistance to pathogen infection in roots (Mäkäräinen et al. 1994) and among potato genotypes reported to have resistance to the formation of root galls at the Columbia Basin (WA) resistant clones were derived from the introgression of S. bulbocastanum and the resistant cultivar Summit Russet appeared more than once in their parental background (Nitzan et al. 2008).
Although the mechanisms involved in resistance to disease caused by S. subterranea are poorly studied, evidence supports the hypothesis that root and tuber resistance to the disease is under control of multiple genes (Bittara et al. 2016; Falloon et al. 2003; Merz et al. 2012). In addition, disease resistance is inherited in an additive manner (Wastie et al. 1988) and is stable across environments (Bittara et al. 2016; Nitzan et al. 2010).
Management
Management of powdery scab is particularly difficult, and no single strategy currently controls powdery scab or root gall formation effectively, making an integrated approach essential (Falloon 2008). Disease avoidance using uncontaminated seed in uninfested soil represents the best method of disease prevention.
The relative importance of soil inoculum level in causing disease on tubers was conclusively demonstrated by Brierley et al. (2013) who showed that when arbitrary soil inoculum threshold values of 0, <10 and >10 sporosori/g soil were set, it was observed that the number of crops developing powdery scab increased with the level of inoculum quantified in the field soil preplanting.
In field trials carried out to investigate the link between the amount of inoculum added to the soil and disease development, disease incidence and severity on progeny tubers was found to be significantly (P < 0.01) greater in plots with increasing levels of inoculum.
This information allows disease risk assessments to be made by taking soil inoculum concentration, in combination with other biotic and abiotic factors, into account. The use of host resistance represents the most effective, sustainable and cost- effective approach for disease management; in addition, genetic resistance is especially suitable on pathogen populations with low genetic diversity (Gau et al. 2013).
In a study performed across five European countries, no evidence of genotype x pathogen interaction was observed as disease development as was similar for all locations (Merz et al. 2012).
Nevertheless, the use of host resistance is affected by factors defined by the consumer market (Harrison et al. 1997). Resting spores produced by the pathogen are highly resistant to environmental stresses and can remain viable for >10 years (Merz and Falloon 2009), therefore up to 7 years of crop rotation or more that excludes potatoes might be required to reduce the risk of disease development once the pathogen has stablished in the field (Sparrow et al. 2015).
Crop rotation may also have an influence on S. subterranea soil infestation as detectable inoculum was shown to be greater following a potato/ wheat rotation compared to a potato/pea rotation (Shah et al. 2014). Additionally, a study by Qu and Christ (2006), found four of 16 crop and weed species infected with S. subterranea subsequently produced root galls, with sporosori detected on three host species (yellow mustard, oats and tomato).
Similarly, Shah et al. (2010) confirmed the presence of sporosori in root galls formed on the solanaceous weeds hairy and black nightshade and subsequently demonstrated pathogenicity on potato with sporosorus inoculum derived from hairy nightshade. It is therefore possible for S. subterranea to complete its lifecycle in the absence of potatoes.
It is broadly accepted that high soil water content encourages zoospore release and that soil in which most pore spaces are filled with water facilitates movement of zoospores towards the host, and subsequent infection and disease development.
This is supported by associations between seasons of high (or low) rainfall or irrigation treatments with high (or low) levels of powdery scab. The chemical management of powdery scab is difficult and, in some cases, economically unsuitable. The use of seed and soil-applied chemicals to control powdery scab was previously reviewed in some detail by Merz and Falloon (2009).
Adolf B. et al. (2020) Fungal, Oomycete, and Plasmodiophorid Diseases of Potato. In: Campos H., Ortiz O. (eds) The Potato Crop. Springer, Cham
