Potato Wart: A Persistent Soil Borne Threat to Global Potato Production
Potato wart, also known as potato wart disease, black wart, black scab, cauliflower disease, potato tumor, potato cancer, potato canker or warty disease is a devastating fungal infection primarily affecting cultivated potatoes (Solanum tuberosum). The disease is characterized by the development of irregular, cauliflower like galls or warts on tubers, stolones and underground stems. These abnormal growths result from hypertrophy (cell enlargement) and hyperplasia (cell proliferation) induced in host tissues by the pathogen.
The causal organism is Synchytrium endobioticum an obligate biotrophic chytrid fungus that depends entirely on living host cells for survival and reproduction making it impossible to grow in artificial culture media. This pathogen is considered one of the most serious quarantine diseases of potatoes worldwide due to its extreme persistence in soil, where resting spores can remain viable for 30–50 years or longer rendering infested fields unsuitable for potato cultivation for decades.
Its quarantine status arises from the potential for complete yield losses reaching up to 100% in severe cases as infected tubers become deformed, spongy and unmarketable, often being entirely replaced by wart like galls. Although S. endobioticum poses no direct risk to human health, it triggers stringent regulatory measures including zero tolerance policies, field quarantines and trade restrictions. These measures can result in indirect economic losses that far exceed direct yield impacts for example an estimated USD 30 million loss to the economy of Prince Edward Island in 2000 following a single detection.
The disease has major implications for seed potato trade because infected seed tubers serve as the primary pathway for long distance spread. Consequently, many countries enforce strict certification programs and import restrictions to prevent pathogen introduction. Globally, potatoes serve as a staple food for over one billion people and play a crucial role in food security across diverse agroecological regions. Therefore, potato wart represents a significant threat to sustainable potato production by contaminating soils and disrupting international supply chains.
The pathogen is classified as an A2 quarantine pest by the European and Mediterranean Plant Protection Organization (EPPO), designated as a Select Agent by the United States Department of Agriculture (USDA) and the U.S. Department of Health and Human Services (HHS) and listed as a harmful organism in 99 countries underscoring its significant regulatory and economic impact worldwide.
Effective management relies primarily on resistant potato varieties and strict phytosanitary measures, as chemical eradication is impractical due to the pathogen’s soil borne nature and the absence of effective fungicidal control options.

Severe Potato Wart Symptoms Affecting Tuber Development
Historical Background and Origin of Potato Wart Disease
Potato wart disease is believed to have originated in the Andean region of South America, particularly in the high-altitude areas of Peru, Bolivia and Ecuador, where it co-evolved with wild and cultivated potato species (Solanum spp.) over thousands of years. The pathogen likely developed alongside potato domestication, which began approximately 8,000–10,000 years ago. Within its native range the disease existed in relative ecological balance with locally adapted and resistant host populations resulting in limited economic impact.
The global dissemination of the disease began during the mid 19th century following the Irish potato famine (1845–1849), caused by Phytophthora infestans. The crisis prompted extensive importation of Andean potato germplasm into Europe for breeding resistant varieties. It is widely believed that infected tubers or contaminated soil accompanied these introductions allowing the pathogen to enter non-native agricultural systems.
The first recorded observations of potato wart in Europe occurred in the late 1870s in England with early reports from the Liverpool district prior to 1893 and confirmed introductions between 1876 and 1878. In 1887, Hungarian scientist Károly Schilberszky described the pathogen in Hornany (now in Slovakia), initially naming it Chrysophlyctis endobiotica after several years of study. The organism was later reclassified as Synchytrium endobioticum in 1910 by John Percival.
Following its establishment the disease spread rapidly across Europe. It was reported in Czechoslovakia in 1888, Germany and Ireland by 1908, followed shortly by Scotland and Wales and reached the Netherlands around 1914. The spread accelerated during World Wars I and II, when food shortages encouraged the movement and planting of infected seed potatoes, particularly from the United Kingdom facilitating continent wide dissemination by the 1940s.
From Europe, potato wart reached North America, where it was first detected in Newfoundland, Canada in 1909 likely through imports of infected seed potatoes from the United Kingdom. In the United States, the disease was identified in Pennsylvania in 1918 and later in Maryland and West Virginia, prompting strict phytosanitary regulations including embargo measures on potatoes from wart affected countries. The pathogen subsequently spread to other regions, including South Africa and New Zealand via Irish seed trade and was reported in India in 1952 following the introduction of Danish seed potatoes.
In Canada, the disease persisted in Newfoundland and later appeared on Prince Edward Island around 1900 possibly originating from infected backyard garden plantings prior to formal quarantine measures implemented in 1912. The disease caused significant trade disruptions as recently as 2000 due to quarantine restrictions. Although eradication programs successfully eliminated potato wart from several U.S. states by the mid 20th century the pathogen remains a long-term threat because its resting spores can survive in soil for decades.
Today, statutory quarantine regulations, resistant cultivar development and strict phytosanitary measures have limited further spread. Nevertheless, historical trade movements and human mediated transport continue to explain the disease’s uneven and patchy global distribution.
Causal Organism and Taxonomic Classification of Synchytrium endobioticum
Synchytrium endobioticum the causal organism of potato wart disease is classified within the Kingdom Fungi, phylum Chytridiomycota a basal fungal lineage characterized by motile, flagellated zoospores and the absence of true hyphae. It belongs to the class Chytridiomycetes, order Chytridiales (although strong molecular evidence supports its reassignment to the order Synchytriales), family Synchytriaceae, genus Synchytrium and species endobioticum.
The genus Synchytrium comprises approximately 200 species of endobiotic (intracellular) and holocarpic parasites meaning the entire thallus converts into reproductive structures. These fungi produce inoperculate sporangia and primarily infect higher plants. Among them, S. endobioticum is the most economically significant species due to its strict host specificity to potato (Solanum tuberosum).
Traditionally, the pathogen was placed in the subgenus Mesochytrium; however, recent studies based on germination behavior and molecular characteristics suggest a possible reclassification into Microsynchytrium. As an obligate biotrophic parasite, S. endobioticum depends entirely on living host cells for nutrition and lacks several independent metabolic pathways including those involved in guanosine triphosphate (GTP) synthesis. Unlike many fungi, it does not form mycelium or hyphae.
Instead, the pathogen produces motile haploid zoospores measuring approximately 1.5–3.0 µm in diameter which are responsible for host infection. For long-term survival, it forms thick-walled resting spores (also called winter sporangia), which are golden brown, ridged structures measuring about 35–80 µm in diameter and capable of persisting in soil for decades.
The life cycle includes both asexual and presumed sexual phases. During the asexual stage, summer sporangia release approximately 200–300 zoospores, enabling rapid secondary infection under favorable conditions. Sexual reproduction is believed to involve zygote formation that subsequently develops into resting spores. Infection primarily occurs in actively dividing meristematic tissues under cool and moist environmental conditions, typically at soil temperatures of 10–18°C with high soil moisture.
Genomic analyses indicate that the pathogen possesses a compact genome encoding effector proteins, such as AvrSen1, which facilitate host manipulation and disease development.
Pathotype diversity in S. endobioticum is extensive with more than 40 pathotypes identified worldwide based on virulence patterns against differential potato cultivars. These pathotypes are distinguished using standardized bioassays including the Spieckermann and Glynne–Lemmerzahl methods. Pathotype 1(D1) remains the most widespread and historically dominant form; however, more aggressive pathotypes such as 2(G1), 6(O1), 8(F1), 18(T1), 38(Nevşehir) and 39(P1) have emerged particularly across Europe, where at least 38 pathotypes have been documented.
Many of these newer variants can overcome single gene resistance sources such as Sen1 largely due to mutation, adaptation and possible genetic recombination. This high variability complicates breeding programs and quarantine management as mixed pathotype populations may occur within a single field necessitating continuous monitoring and the deployment of stacked or durable resistance strategies for long-term disease control.
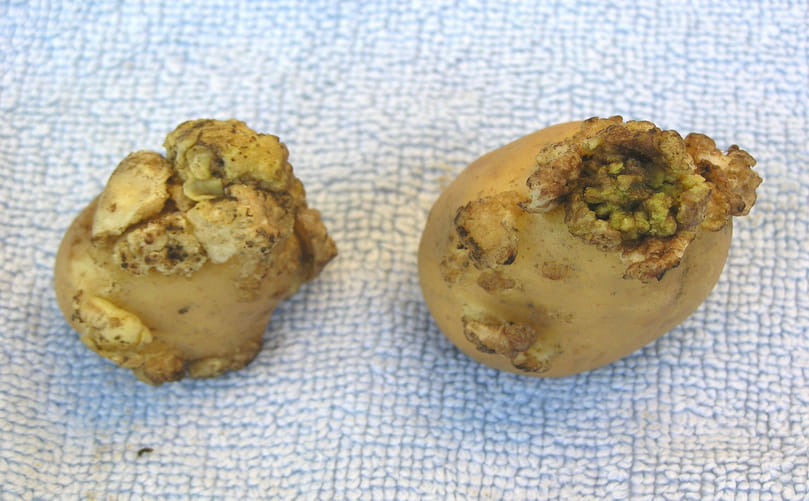
Typical Symptoms of Potato Wart Disease in Solanum tuberosum Tubers
Pathotype Diversity and Host Specificity of Synchytrium endobioticum in Potatoes
The primary pathogen responsible for potato wart disease is Synchytrium endobioticum. No other species within the genus Synchytrium is known to cause comparable symptoms in potatoes or pose a similar level of economic threat. The disease is therefore considered highly species specific, with S. endobioticum recognized as the sole causal agent of potato wart worldwide.
A defining characteristic of S. endobioticum is its extensive pathotype diversity. More than 40 pathotypes have been identified globally based on differences in virulence against a standardized set of differential potato cultivars. Pathotypes are distinguished by their ability to overcome specific host resistance genes making their identification essential for breeding programs, quarantine regulation and disease risk assessment.
Pathotype 1(D1) is the most widespread and historically dominant strain and was the first pathotype described. It occurs globally and has served as the reference pathotype for resistance screening. Other economically important pathotypes include 2(G1) (first identified in Germany), 6(O1) (Austria), 8(F1) and 18(T1) (formerly reported from the German Democratic Republic). These pathotypes are particularly significant in Europe because they infect a wider range of potato cultivars and can overcome resistance effective against pathotype 1(D1).
In recent decades, more aggressive or so-called “higher” pathotypes have emerged. Examples include pathotype 38(Nevşehir), first detected in Turkey in 2005 and pathotype 39(P1), identified in Poland. These variants are frequently associated with cool, humid and mountainous regions such as the Alps and Carpathians, where environmental conditions favor disease persistence and evolution. Their emergence has challenged previously resistant potato varieties and increased concerns regarding long distance spread through trade and soil movement.
Pathotype identification relies primarily on standardized bioassays using differential host cultivars. Two widely accepted methods are the Glynne–Lemmerzahl method, which uses controlled pot tests with infested soil and differential cultivars and the Spieckermann method based on soil infestation assays. These procedures recommended under EPPO diagnostic protocols determine virulence by evaluating wart formation on specific differential varieties, such as ‘Deodara’ for pathotype 1(D1), ‘Producent’ for 2(G1) and ‘Talent’ for 18(T1).
Molecular tools have significantly improved pathotype differentiation. Techniques such as simple sequence repeat (SSR) marker analysis have revealed at least three major genetic clusters among European and North American populations suggesting limited historical introductions followed by regional evolution, recombination and human mediated dispersal. Comparative genomic studies of isolates such as MB42 (pathotype 1(D1)) and LEV6574 (pathotype 6(O1)) demonstrate variation in effector genes including mutations in AvrSen1, enabling certain pathotypes to evade host immune responses.
This diversity arises through mutation, potential sexual recombination and adaptive evolution, often resulting in mixed pathotype populations within individual fields. Such complexity complicates disease management and resistance breeding. Single gene resistance sources, such as Sen1 are frequently overcome, highlighting the need to pyramid multiple resistance loci to achieve durable resistance.
Quarantine and regulatory decisions are strongly influenced by pathotype virulence. European and Mediterranean Plant Protection Organization (EPPO) guidelines commonly prioritize detection of the four principal pathotypes 1(D1), 2(G1), 6(O1) and 18(T1) for regulatory monitoring, while detection of higher pathotypes typically triggers stricter containment and phytosanitary measures due to their greater threat to modern cultivars.
The continued emergence of new pathotypes underscores the necessity for ongoing surveillance, standardized diagnostics, resistant cultivar development and international collaboration to limit global spread and protect potato production systems.
Host Range and Susceptible Plant Species of Synchytrium endobioticum
The principal and most economically important host of Synchytrium endobioticum is the cultivated potato (Solanum tuberosum). In this host, the pathogen induces characteristic wart like galls on tubers, stolons and underground stems, resulting in severe yield losses and complete market rejection due to poor tuber quality.
Within its native Andean region, the pathogen is also capable of infecting several wild Solanum species, which are believed to have co-evolved with the fungus and may serve as natural reservoirs and sources of genetic diversity. Reported susceptible wild relatives include Solanum acaule, S. chacoense, S. commersonii, S. demissum, S. jamesii and S. maglia. These wild hosts are of particular epidemiological importance because they can maintain the pathogen in the environment even in the absence of cultivated potatoes. In Mexico and other regions rich in native potato relatives, occasional but largely unconfirmed reports suggest infections in additional wild Solanum species, indicating the possibility of alternative hosts under natural conditions.
Under controlled experimental conditions, the host range appears broader within the family Solanaceae. Artificial inoculation has demonstrated that resting spores may form in several solanaceous plants although infections are typically weak and produce few or no visible symptoms. Such experimentally susceptible plants include tomato (Solanum lycopersicum), where only limited gall formation has been observed as well as Solanum dulcamara (bittersweet nightshade), Solanum nigrum (black nightshade), Solanum villosum, Nicotiana tabacum (tobacco), species of Physalis, Capsicastrum, Datura (e.g., jimsonweed), Hyoscyamus, Lycium, Nicandra physalodes (apple of Peru), Schizanthus and Duboisia. However, natural field infections in these non-potato hosts are extremely rare and they are not considered significant contributors to disease epidemics or major inoculum sources.
The pathogen shows strong tissue specificity infecting actively dividing meristematic tissues such as tuber eyes, stolon tips and sprout buds. Potato roots are not susceptible to infection, restricting disease development primarily to underground organs and under severe conditions occasionally to basal above ground tissues.
No non-solanaceous hosts have been confirmed reinforcing the narrow host specificity of S. endobioticum as an obligate biotrophic parasite dependent on living solanaceous cells. This restricted host range allows partial disease management through crop rotation with non-host species. Nevertheless, eradication remains challenging in regions where wild Solanum populations persist as they may harbor latent infections and sustain long-term soil inoculum.
Resistance breeding frequently utilizes genes derived from wild potato relatives; however, ongoing pathotype evolution can overcome single resistance sources. Consequently, continuous screening of diverse germplasm and the deployment of multiple resistance genes are essential for achieving durable resistance against potato wart disease.

Advanced Potato Wart Infection Showing Tumor like Growth on Tubers
Global Distribution and Spread of Potato Wart Disease
Potato wart disease caused by Synchytrium endobioticum, shows a fragmented but worldwide distribution, largely shaped by historical trade movements, strict quarantine regulations and environmental suitability. The pathogen is considered present on most potato growing continents although typically restricted to localized areas due to intensive phytosanitary control measures.
The disease is believed to have originated in the Andean highlands of South America, particularly regions of present-day Peru and Bolivia, where it co-evolved with wild and cultivated potato species. From this native center the pathogen was introduced into Europe during the late nineteenth century, most likely through infected seed tubers transported for breeding programs following the Irish potato famine. By the early twentieth century, potato wart had spread across several European countries, including Germany, the United Kingdom, the Netherlands, Poland and other central and northern European regions becoming one of the earliest internationally regulated plant diseases.
Today, Europe remains the region with the widest recorded occurrence, although distribution is highly localized and officially controlled. The pathogen occurs in multiple European countries such as Germany, Ireland, Poland, Ukraine, Russia, Turkey and the United Kingdom, usually confined to demarcated infested zones under quarantine restrictions. Similar restricted distributions are reported elsewhere worldwide.
In the Americas, the disease persists in parts of South America within its native range and occurs in Canada, particularly Newfoundland and Prince Edward Island, where it remains under strict regulatory control. Earlier outbreaks detected in the United States during the early twentieth century were successfully eradicated through quarantine and sanitation measures.
Across Asia, confirmed occurrences include India (mainly restricted Himalayan regions), China, Bhutan3 and Nepal, while in Africa it has been reported in South Africa and Tunisia. In Oceania, the pathogen exists in New Zealand under limited distribution. Many countries remain free from the disease due to long standing import restrictions and surveillance programs.
The spread of potato wart disease is overwhelmingly human mediated. Long distance dissemination occurs primarily through infected seed tubers and soil attached to planting material. Local spread results from contaminated machinery, tools, footwear, irrigation runoff, animals fed infected tubers and movement of infested soil. Natural dispersal is limited because motile zoospores travel only a few centimeters in wet soil; however, thick-walled resting spores can survive in soil for decades allowing the pathogen to persist long after initial introduction.
Although international quarantines and trade bans have significantly reduced large scale expansion, periodic re-emergence in previously controlled regions demonstrates that global trade and soil movement continue to pose risks. Consequently, potato wart remains one of the most strictly regulated quarantine diseases worldwide requiring continuous monitoring and coordinated international phytosanitary measures.
Symptoms and Disease Identification of Potato Wart Disease
Symptoms of potato wart disease, caused by Synchytrium endobioticum are primarily restricted to underground plant parts and are characterized by abnormal, cauliflower like warty growths or galls developing on tubers, stolons and underground stems. These structures arise from pathogen-induced hypertrophy (cell enlargement) and hyperplasia (cell proliferation) in infected tissues. Infection typically begins in actively dividing meristematic regions such as tuber eyes, stolon tips, sprout buds and stem bases, while true roots remain unaffected.
Early symptoms appear as soft, pulpy, white to cream colored swellings that closely resemble normal host tissue and are easier to cut than healthy tuber flesh. As the disease progresses, these swellings enlarge into irregular, rough and corrugated masses resembling cauliflower heads. Gall size may vary from small pinhead-like structures to large deformities capable of covering the entire tuber surface. Advanced infections often distort tubers into spongy, irregular shapes that are no longer recognizable as marketable potatoes.
Color changes accompany disease development. Young galls remain white or pale but may turn green when exposed to light due to chlorophyll formation. With age, tissues become brown and eventually black as they degenerate and decay releasing large numbers of resting spores into the surrounding soil. When infection occurs early during tuber development normal tuber formation may be completely suppressed resulting in total yield loss.
Above-ground symptoms are uncommon and generally nonspecific. In heavily infected plants, slight reductions in vigor, distorted shoots or small greenish yellow wart like swellings at the stem base may occasionally be observed. Because visible symptoms often develop late, the disease may remain undetected until harvest or even during storage, where galls can continue enlarging under moist conditions. Consequently, post-harvest inspection plays an important role in disease detection.
Infected tubers are rendered completely unmarketable due to severe deformities, tissue decay and strict quarantine regulations. The spongy gall tissue also becomes highly susceptible to secondary microbial infections, accelerating deterioration.
Field diagnosis is primarily based on visual identification of characteristic wart-like galls on harvested tubers. Definitive confirmation, however, requires laboratory examination. Microscopic observation reveals distinctive resting spores that are solitary, thick walled, golden brown and typically 35–80 µm in diameter providing a key diagnostic feature.
Potato wart disease may be confused with several other disorders. Powdery scab, caused by Spongospora subterranea, produces powdery pustules containing clustered spore balls rather than solid warts. Potato smut (Thecaphora solani) forms dark, powdery masses within tuber tissues. Physiological abnormalities such as bud proliferation (“false wart”) or damage caused by root knot nematodes may also resemble early symptoms but lack true wart structures and resting spores. Accurate diagnosis therefore relies on laboratory confirmation using bioassays or molecular detection methods, including PCR assays targeting ITS or 18S rDNA regions specific to S. endobioticum.

Severe Expression of Potato Wart on Tubers Caused by Synchytrium endobioticum
Disease Cycle and Epidemiology of Potato Wart Disease
The disease cycle of potato wart, caused by Synchytrium endobioticum begins with thick-walled resting spores (also known as winter sporangia) that persist in soil or decomposing wart tissue for exceptionally long periods, often surviving 20–50 years or more. This remarkable longevity enables the pathogen to remain viable even in the prolonged absence of potato cultivation.
Under favorable environmental conditions in spring typically soil temperatures above 8–10°C combined with high soil moisture from rainfall or irrigation and adequate aeration resting spores germinate. Each resting spore releases approximately 200–300 haploid, flagellated zoospores measuring about 1.5–3.0 µm in diameter. These motile zoospores move through water films in the soil and infect susceptible potato tissues, primarily targeting actively dividing meristematic regions such as tuber eyes, stolon tips, sprouts and stem bases.
Following penetration of host epidermal cells, the pathogen develops into thin-walled summer sporangia within infected tissue. These structures produce additional zoospores that initiate repeated secondary infection cycles. Under cool and moist conditions, infection cycles may recur every 10–12 days during the growing season, resulting in rapid enlargement of wart tissue, which functions as a strong nutrient sink and can expand dramatically within a short period.
When environmental conditions become unfavorable, particularly during soil drying or plant senescence, zoospores may fuse pairwise to form zygotes, representing a presumed sexual stage of the life cycle. These zygotes develop into new thick-walled resting spores inside host tissues. As infected galls mature and decay in the field or during storage resting spores are released back into the soil thereby completing the disease cycle.
Because S. endobioticum is an obligate biotrophic pathogen, it cannot be cultured on artificial media, which limits experimental study and complicates epidemiological research. Although only a small proportion of resting spores germinate in a given season (often around 10%), their extremely high survival capacity ensures long-term persistence and contributes to irregular patchy disease outbreaks.
From an epidemiological perspective, disease spread is predominantly human mediated. The most important pathway is the movement of infected seed tubers including those carrying latent infections or contaminated soil. Additional routes include contaminated agricultural machinery, tools, footwear, irrigation water and manure derived from animals fed infected potatoes. Natural spread is comparatively limited and occurs mainly through short distance movement of infested soil via water erosion, wind-blown particles or soil organisms such as earthworms.
Continuous potato cultivation promotes inoculum buildup and increases disease severity, whereas rotation with non-host crops can gradually reduce viable spore populations, sometimes by 70–99% over extended periods. However, eradication is rarely achieved because resting spores remain viable for decades and wild Solanum species may serve as reservoirs maintaining low pathogen populations.
Pathotype diversity further influences epidemiology as more aggressive variants often emerge or persist in cool, humid and mountainous environments favorable to disease development. Modern molecular approaches including mitochondrial genome analysis and population genetics studies have improved understanding of global pathogen dissemination patterns originating from the Andean center of origin.
The extraordinary soil persistence of resting spores combined with the absence of effective curative treatments makes potato wart one of the most serious quarantine diseases of potato worldwide. Outbreaks frequently remain undetected until harvest, emphasizing the importance of surveillance, certified seed systems and strict phytosanitary management.
Factors Influencing Disease Severity of Potato Wart
Disease severity in potato wart, caused by Synchytrium endobioticum, is determined by a complex interaction among environmental conditions, host susceptibility, pathogen characteristics and agronomic practices. Among these factors, cool and moist soil conditions are the most critical drivers promoting infection, pathogen development and gall formation.
Environmental conditions strongly influence disease expression. Optimal soil temperatures for resting spore germination and zoospore activity generally range from 8–18°C with infection occurring most readily between approximately 12–18°C under moist conditions. Regions characterized by cool summers, prolonged soil moisture and cold winters favor disease persistence and epidemic development. High annual rainfall, typically around 700–800 mm or more, combined with poor soil drainage or excessive irrigation during tuber initiation, significantly increases disease severity by enhancing zoospore movement and repeated infection cycles. Under such conditions, gall development intensifies and yield losses in susceptible cultivars may reach 100%.
Soil moisture is particularly important because zoospores require free water films for movement and host infection. Waterlogged or compacted soils therefore promote disease, whereas well drained soils reduce infection pressure. Soil pH has a relatively minor influence as the pathogen can survive across a wide range (approximately pH 3.9–8.5) although slightly acidic soils may support better spore survival. Adequate soil oxygen and the presence of nitrates are also known to stimulate resting spore germination.
Host genotype is another decisive factor. Potato cultivars lacking effective resistance genes are highly susceptible and develop severe wart formation and tuber deformation. Resistant varieties carrying resistance genes such as Sen1 and related loci can restrict infection; however, disease severity increases when pathogen pathotypes capable of overcoming these resistances are present. Aggressive pathotypes including variants such as 18(T1) or 38(Nevşehir) can infect previously resistant cultivars leading to renewed outbreaks.
Disease intensity is also closely linked to soil inoculum levels. Fields with high concentrations of resting spores, resulting from previous infestations or repeated potato cultivation, exhibit greater disease incidence and severity. Continuous potato cropping or short crop rotations accelerate inoculum buildup, whereas long rotations with non-host crops gradually reduce viable spore populations.
Pathogen-related factors further influence disease outcomes. The existence of more than 40 pathotypes with varying virulence contributes to differences in disease severity across regions. Mixed pathotype populations within a single field may accelerate resistance breakdown and complicate control measures.
Agronomic practices can either mitigate or intensify disease expression. Heavy soils that retain moisture, deep planting that prolongs exposure of developing tissues to infested soil and early season infections affecting sprouts can reduce plant emergence and substantially increase yield losses.
Overall, severe potato wart development is most commonly associated with cool, wet soils in northern or mountainous potato growing regions, combined with susceptible cultivars, high inoculum density and aggressive pathogen pathotypes. Effective management therefore requires an integrated approach combining resistant varieties, crop rotation, soil hygiene and strict phytosanitary measures.
Global Economic Burden and Quality Losses Caused by Potato Wart Disease
Potato wart disease caused by Synchytrium endobioticum, imposes substantial global economic losses through a combination of direct yield reductions, severe quality deterioration and extensive indirect costs associated with quarantine enforcement, trade restrictions and long-term land-use limitations. Although strict phytosanitary measures have reduced widespread epidemics in many regions the disease remains one of the most economically consequential quarantine pathogens affecting potato production worldwide.
Direct economic impacts arise primarily from yield loss and tuber destruction. In moderately infested fields, yield reductions may reach approximately 50%, while severe infestations can result in total crop failure when developing tubers are completely replaced by wart tissue. Infected tubers become highly deformed, spongy and susceptible to rapid decay and secondary microbial infections. As a result, they are unsuitable for fresh markets, processing industries or use as seed potatoes. Even when infection levels are relatively low, affected lots are rejected under phytosanitary regulations causing immediate financial losses. Post-harvest losses may further increase because wart tissues can continue expanding under moist storage conditions.
Indirect economic impacts often exceed direct yield losses. Once detected, infested fields are typically placed under long-term quarantine because resting spores can survive in soil for decades, frequently restricting potato cultivation for 20–50 years or longer. Farmers may be forced to switch to less profitable crops or abandon production entirely, resulting in significant opportunity costs. Governments and regulatory agencies also incur expenses related to surveillance, diagnostics, containment programs, compensation schemes and enforcement of phytosanitary regulations.
International trade disruptions represent one of the most significant economic consequences of potato wart outbreaks. Detection of the pathogen can trigger immediate export suspensions and market closures. A well-known example occurred in Prince Edward Island, Canada, where a potato wart detection led to export restrictions and economic losses estimated at approximately USD 30 million within the first year due to disrupted trade and surplus management costs. Because potatoes represent a major global commodity valued in billions of dollars annually, the introduction of the disease into currently disease-free regions could have severe economic repercussions. For example, in countries heavily dependent on potato exports, even temporary trade bans could affect a substantial portion of national agricultural revenue and rural employment.
Historical evidence highlights the long-term economic implications of the disease. Early twentieth century trade embargoes on infected regions significantly reduced international potato exports, while more recent outbreaks in parts of Europe and Asia have required extensive containment measures and resulted in large areas of agricultural land being removed from potato production. Regulatory responses including crop destruction, movement restrictions and prolonged monitoring programs, further increase economic costs for both growers and governments.
Beyond direct farm level losses potato wart disease disrupts seed potato supply chains, limits breeding and germplasm exchange and creates uncertainty in international markets. Regions currently free from the disease invest heavily in preventive measures because maintaining pest free status protects market access and industry stability.
Overall, while strict quarantine systems have helped limit widespread yield losses in many countries the extraordinary persistence of S. endobioticum in soil and its quarantine status generate long-lasting economic consequences. The combined effects of production losses, land-use restrictions, regulatory compliance and trade barriers underscore the importance of continuous surveillance, resistant cultivar development and coordinated international management to safeguard global potato production systems.
Management and Control Strategies for Potato Wart Disease
Effective management of potato wart disease, caused by Synchytrium endobioticum, relies on an integrated strategy centered on prevention, exclusion and strict containment. Because the pathogen produces long lived resting spores capable of surviving for decades in soil and no curative treatment exists, eradication is extremely difficult once infestation occurs. Consequently, successful control depends on combining resistant cultivars, phytosanitary measures, cultural practices and regulatory enforcement rather than relying on chemical interventions.
Historically, chemical control methods such as mercury compounds, sulfur, copper formulations, chlorine-based disinfectants and formaldehyde were tested but proved ineffective at eliminating resting spores and caused severe environmental and soil damage. These approaches were therefore abandoned. Modern soil fumigants such as metham sodium may provide limited suppression under specific conditions but are not considered practical or sustainable management options. Biological approaches including chitin amendments, urea applications, organic fertilizers or crab shell meal, have shown experimental potential for reducing spore activity; however, results remain inconsistent and these methods are not yet reliable for field scale disease control. As a result, management programs primarily emphasize prevention and regulatory containment.
Resistant Varieties: The cultivation of resistant potato varieties represents the most effective and environmentally sustainable strategy for managing potato wart disease. Resistant cultivars prevent pathogen establishment within host tissues, thereby limiting gall formation and reducing inoculum buildup in soil.
Resistance breeding programs have incorporated resistance genes such as Sen1–Sen5, many of which originate from wild Solanum species including S. acaule and S. vernei. Modern breeding increasingly uses marker assisted selection and genomic tools to combine multiple resistance loci, improving durability against diverse pathogen pathotypes. Early breeding efforts successfully produced cultivars resistant to the historically dominant pathotype 1(D1); however, the emergence of more aggressive pathotypes, such as 38 (Nevşehir) has required continued breeding efforts and resistance diversification.
Resistance screening is conducted using standardized bioassays such as the Glynne-Lemmerzahl and Spieckermann methods along with national testing protocols to verify cultivar performance under controlled infection conditions. In many countries, resistant varieties are mandatory within buffer zones surrounding infested fields allowing limited potato production while minimizing pathogen spread. Nevertheless, resistance breakdown remains a risk when single resistance genes are deployed making gene stacking and international breeding collaboration essential for long-term control.
Phytosanitary Measures: Phytosanitary practices are critical for preventing introduction and dissemination of the pathogen. The use of certified, disease-free seed potatoes is mandatory in most production systems, ensuring planting material originates from non-infested areas and is free from contaminated soil.
Strict sanitation protocols are required for agricultural machinery, tools, vehicles and footwear to prevent movement of infested soil. Cleaning and disinfection using approved agents, such as bleach solutions or quaternary ammonium compounds, significantly reduce transmission risk. Movement of soil, rooted plants and agricultural waste from infested zones is heavily restricted. Manure derived from animals fed infected potatoes must be properly composted or avoided as resting spores can survive passage through digestive systems.
Processing regulations may require steaming, incineration or chemical treatment of infected tubers to destroy viable spores. International and regional phytosanitary organizations establish guidelines to maintain disease-free status and prevent cross border spread.
Cultural Practices: Cultural management practices aim to reduce inoculum levels and minimize environmental conditions favorable for infection. Long-term crop rotation with non-host species such as cereals, maize or brassicas can gradually reduce viable resting spore populations although complete elimination is unlikely due to the pathogen’s longevity. Avoiding continuous potato cultivation is essential to prevent inoculum buildup.
Field selection also influences disease risk. Well drained soils reduce water films necessary for zoospore movement, whereas heavy or poorly drained soils promote infection. Practices that limit soil erosion including cover cropping and careful water management help reduce local dispersal of contaminated soil.
Experimental soil amendments, including chitin or nitrogen-based treatments may suppress spore germination but should be considered supportive rather than primary management tools. Regular field surveys and early detection programs are vital for rapid response and containment.
Regulatory and Quarantine Measures: Regulatory control forms the backbone of potato wart management worldwide. Synchytrium endobioticum is classified as a quarantine pathogen in many countries and strict zero tolerance policies are enforced following detection.
Infested fields are typically placed under long-term quarantine, often lasting at least 20 years during which potato cultivation is prohibited. Buffer zones may permit only resistant varieties under controlled conditions. Infected crops are destroyed through approved methods such as herbicide desiccation, steaming or incineration to prevent further spore release.
Movement restrictions apply to potatoes, soil, machinery and plant materials originating from infested areas. National management programs outline surveillance, testing and containment procedures designed to prevent spread and protect export markets. International organizations provide harmonized guidelines and fields are only released from quarantine after extended monitoring confirms the absence or non-viability of resting spores.
Overall, integrated management combining resistant cultivars, rigorous sanitation, informed cultural practices and strict regulatory oversight remain the only effective strategy for limiting the long-term impact of potato wart disease and safeguarding global potato production systems.
Management Challenges in Controlling Potato Wart Disease
Management of potato wart disease caused by Synchytrium endobioticum, presents significant challenges due to the pathogen’s unique biological characteristics, evolving virulence and strict regulatory requirements. These factors collectively make long-term control difficult, costly and often economically burdensome for growers and regulatory authorities.
One of the greatest challenges is the extraordinary longevity of resting spores, which can remain viable in soil for more than 40–50 years even in the absence of potato cultivation. This persistence renders infested land unsuitable for potato production for decades and makes eradication virtually impossible once the pathogen becomes established. Resting spores are highly resistant to environmental stresses, surviving desiccation, freezing temperatures and many chemical treatments and they may persist in soil layers reaching depths of up to approximately 50 cm.
The absence of effective chemical control options further complicates management. Earlier attempts using mercury, sulfur, copper compounds, chlorine-based disinfectants and formaldehyde failed to eliminate the pathogen and caused severe environmental damage leading to their discontinuation. Modern fumigants such as metham sodium provide only partial suppression and are generally considered impractical because of environmental risks, high costs and inconsistent performance. Biological amendments including chitin-based materials or urea treatments have demonstrated potential under experimental conditions but show variable and unreliable results under field conditions limiting their practical application.
Another major obstacle is the emergence and diversification of pathogen pathotypes. More than 40 pathotypes have been identified worldwide, many capable of overcoming resistance genes deployed in cultivated potato varieties. Regions with intensive potato monoculture and short crop rotations, such as starch potato production systems create strong selection pressure that favors pathogen adaptation. The appearance of aggressive pathotypes, including variants capable of overcoming resistance previously effective against pathotypes 1(D1), 2(G1), 6(O1) and 18(T1), illustrates the limitations of relying on single resistance genes and highlights the need for durable, multi-gene resistance strategies.
Early disease detection also remains problematic. Symptoms are largely confined to underground plant parts and often become visible only at harvest, allowing the pathogen to remain undetected for extended periods. Latent infections and mixed pathotype populations within fields further complicate diagnosis and accurate assessment of virulence delaying management responses.
Economic and regulatory pressures add additional complexity. Detection of potato wart typically triggers strict quarantine measures including long-term land use restrictions that may exceed 20 years, movement controls and mandatory destruction of infected crops. These measures can result in substantial financial losses for growers and regional agricultural economies due to lost production opportunities, increased compliance costs and restricted market access. Regulatory restrictions may also limit researcher access to infested sites, slowing progress in epidemiological studies and development of improved control methods.
Breeding resistant cultivars presents further challenges because potato is genetically complex and resistance traits must be combined with desirable agronomic characteristics such as yield, processing quality and storage performance. This makes resistance development time consuming and resource intensive.
Finally, increasing global trade and movement of agricultural materials elevate the risk of pathogen dissemination across borders, while climate variability may expand regions suitable for disease establishment. Together, these biological, economic and regulatory constraints underscore the need for adaptive, integrated management strategies combining resistant varieties, strict phytosanitary systems, continuous monitoring and international cooperation to effectively mitigate potato wart disease.
Integrated Detection and Diagnostic Approaches for Potato Wart Disease
Detection and diagnosis of potato wart disease rely on a combination of visual assessment, microscopic examination, molecular techniques and bioassays to identify symptoms, confirm the presence of the pathogen, quantify inoculum levels and determine pathotypes, thereby supporting accurate quarantine and regulatory responses. Field diagnosis begins with inspection of tubers, stolons and underground stems for characteristic cauliflower-like galls that initially appear white and later turn dark brown to black as they decay. However, these symptoms may be confused with diseases such as powdery scab or potato smut making laboratory confirmation essential.
Soil testing commonly involves wet sieving or dry sieving methods to extract resting spores followed by chloroform flotation and centrifugation to concentrate spores for microscopic observation. Under light microscopy, the pathogen is identified by its distinctive golden brown, thick-walled resting spores measuring approximately 35–80 μm in diameter. To assess spore viability, bioassays such as the tomato bait plant test or the Spieckermann pot test are conducted using susceptible host plants, where wart formation confirms infectivity after approximately 6–8 weeks.
Molecular diagnostic methods have significantly improved detection accuracy. Real time PCR assays targeting ITS or 18S rDNA regions provide high sensitivity and specificity, enabling detection of as few as 1–3 spores per gram of soil. Quantitative PCR (qPCR) further allows estimation of inoculum density, while emerging viability assays evaluate metabolic activity or DNA integrity to differentiate live spores from non-viable ones. Pathotype identification primarily relies on standardized bioassays including the Glynne–Lemmerzahl and Spieckermann methods in which differential potato cultivars are inoculated to observe virulence reactions; however, these procedures are time intensive and may require up to three months for completion.
Recent diagnostic innovations include gas chromatography mass spectrometry (GC-MS) for profiling volatile organic compounds associated with infection, offering potential for rapid and non-invasive detection. Reliable diagnosis also depends on proper soil sampling strategies, such as tare or grid sampling methods to ensure representative analysis. Typically, confirmation requires detection of at least two resting spores along with molecular verification.
These integrated approaches are incorporated into international diagnostic protocols to ensure reliability although challenges such as low spore germination rates and complex sample preparation remain highlighting the need for combined diagnostic strategies to achieve accurate disease identification.
Regulatory Status and Quarantine Significance of Potato Wart Disease
Potato wart is internationally recognized as a high priority quarantine disease because of its severe economic consequences, long-term soil persistence and capacity to disrupt global potato trade. The causal pathogen, Synchytrium endobioticum is classified as an A2 quarantine pest by the European and Mediterranean Plant Protection Organization (EPPO) and is regulated as a Select Agent by both the United States Department of Agriculture (USDA) and the U.S. Department of Health and Human Services (HHS). Owing to its global phytosanitary importance, the pathogen is regulated in more than 90 countries under strict zero tolerance policies, reflecting its status as one of the most significant quarantine pathogens affecting potatoes worldwide.
Within the European Union, legislative frameworks such as Directives 69/464/EEC and 2000/29/EC mandate strict containment measures, including prohibition of potato cultivation and other transplantable crops in infested plots until official clearance is granted. Similar regulatory rigor exists in Canada, where the disease is governed under the Plant Protection Act, triggering immediate land use restrictions, movement controls, sanitation protocols and long-term monitoring programs following detection. A notable example occurred in Prince Edward Island, where confirmed outbreaks resulted in extensive quarantine zones and prolonged management strategies to protect export markets.
The quarantine importance of potato wart primarily arises from the extraordinary longevity of resting spores, which can remain viable in soil for more than 40 years. As a result, infested land may become unsuitable for potato production for decades causing indirect economic losses that often exceed direct yield damage. Historical detections in the United States led to trade embargoes and strict phytosanitary enforcement, while modern regulations commonly impose quarantine periods of 20 years or more with buffer zones allowing cultivation only of resistant varieties.
Global phytosanitary legislation developed over the past several decades includes mandatory phytosanitary certification, import restrictions from infested regions and rigorous de-scheduling procedures requiring long-term confirmation of spore non-viability, often extending from 10 to over 20 years. EPPO Standard PM9/005 provides guidance for surveillance, containment and suppression strategies, emphasizing scheduled fields without potato cultivation and controlled safety zones. Although these regulatory measures effectively limit pathogen spread, they also impose substantial economic and logistical burdens on growers and national industries, highlighting the critical role of quarantine systems in protecting international potato trade and ensuring long-term food security.
Research Advances and Future Directions in Potato Wart Disease Management
Recent research on potato wart disease has increasingly integrated genomics, molecular diagnostics and microbiome science to improve detection, resistance breeding and long-term suppression strategies, supporting more sustainable management under evolving pathogen populations and changing climatic conditions. Advances in genome sequencing have generated reference genomes for multiple Synchytrium endobioticum isolates, enabling detailed analysis of effector diversity and pathogenicity mechanisms. Variations in effector genes, such as mutations associated with AvrSen1 have been linked to the emergence of new pathotypes capable of overcoming host resistance, while comparative genomic studies have helped clarify evolutionary relationships and potential migration pathways of the pathogen.
Molecular characterization has also progressed through the development of highly informative genetic markers. Simple sequence repeat (SSR) markers, including panels comprising dozens of loci and numerous alleles have enabled clustering of pathogen isolates into distinct genetic groups, improving understanding of population structure, virulence evolution and regional spread patterns. These insights directly support surveillance programs and resistance deployment strategies.
Resistance breeding has advanced significantly with the identification and mapping of major wart resistance genes (Sen1–Sen5), which function as TNL-type immune receptors in potato. Marker assisted selection now accelerates the incorporation of resistance into elite cultivars, while modern genome editing technologies such as CRISPR–Cas9 and TALENs are being explored to stack multiple resistance genes and enhance durability. Current breeding objectives increasingly integrate wart resistance with agronomic performance traits including yield stability, processing quality and nutritional value.
Microbiome research represents another emerging frontier. High resolution profiling using full length 16S rRNA sequencing platforms, including nanopore-based technologies has revealed microbial communities associated with disease suppressive soils. Functional analyses suggest that specific microbial networks may inhibit pathogen development providing a foundation for hypothesis driven development of biological control agents. Although promising, these findings require extensive field validation before practical application.
Diagnostic innovation continues to improve early detection and risk assessment. Standardized quantitative PCR (qPCR) assays now enable sensitive detection and approximate quantification of pathogen DNA, while research into viability testing aims to distinguish living from non-viable spores. Complementary approaches such as volatile organic compound profiling using gas chromatography mass spectrometry (GC–MS) are being investigated as rapid, non-invasive detection tools. Enhanced bioassay systems are also under development to shorten evaluation time for spore infectivity.
Suppression research is exploring environmentally compatible approaches including biopesticides, phosphite-based treatments and organic amendments such as chitin, which may reduce spore germination or pathogen activity under certain conditions. However, field consistency remains a key challenge highlighting the need for integrated management frameworks rather than single solution approaches.
Future research directions emphasize stronger international collaboration to standardize spore viability assessment and quarantine thresholds, alongside the adoption of advanced breeding strategies such as hybrid breeding systems for faster resistance introgression. Long-term microbiome studies conducted under field conditions are expected to validate biological control concepts, while multi-omics investigations combining genomics, transcriptomics and metabolomics aim to clarify unresolved aspects of pathogen biology including infection triggers and reproductive processes. Collectively, these advances are expected to support durable disease containment, reduce quarantine impacts and improve resilience of global potato production systems under increasing trade and climate pressures.

